2016 09 29 Cochlear launches the worlds thinnest full length perimodiolar electrode
Renee Oehlerking
Cochlear Americas
303-264-2104
roehlerking@Cochlear.com
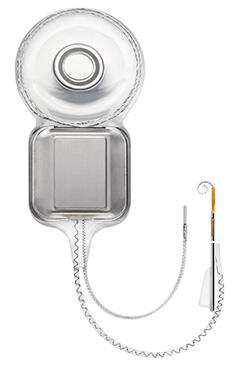
The new Cochlear™ Nucleus® Profile Implant with Slim Modiolar Electrode (CI532) is designed to preserve hearing structures, boost hearing performance and is easy for surgeons to use
Centennial, Colo., (September 29, 2016) — Cochlear Limited (ASX: COH), the global leader in implantable hearing solutions, announces today the launch of the Cochlear Nucleus Profile Implant with Slim Modiolar Electrode (CI532).
The all new Slim Modiolar Electrode (CI532) is the world’s thinnest full-length perimodiolar electrode and comes on the most reliable implant (Nucleus Profile) available on the market today.1,2 The new electrode sits close to the hearing nerve, and its thin design is intended to protect and preserve the delicate structures of the inner ear in order to deliver the best hearing performance possible.3,4
Independent research shows electrode contacts positioned closer to the hearing nerve significantly improve hearing performance in patients with severe to profound hearing loss.5-8
“Our new Slim Modiolar Electrode is the latest perimodiolar array innovation in our portfolio of electrodes, and it really does make a difference for our customers and the surgeon,” said Tony Manna, President of Cochlear North America. “Feedback from leading surgeons around the world provided us with valuable insights as we developed this electrode. Cochlear now offers the world’s thinnest, full-length perimodiolar electrode, offering smooth and easy insertion during surgery and optimal hearing for our customer afterwards.”
The Slim Modiolar Electrode features a unique insertion mechanism that allows surgeons to tailor their electrode insertion approach for the individual patient’s needs, making insertion smooth and easy.9,10 It is compatible with round window, extended round window and cochleostomy insertion approaches and can be reloaded if required.
The addition of the Slim Modiolar Electrode further strengthens Cochlear’s market-leading portfolio of electrodes uniquely designed for preservation, performance and surgeon preference. The Nucleus implant portfolio offers surgeons and their patients a wide choice of electrodes for different types of hearing loss.
About Cochlear Limited (ASX: COH)
Cochlear is the global leader in implantable hearing solutions. The company has a global workforce of 2,800 people and invests more than AUS$100 million a year in research and development. Products include cochlear implants, bone conduction and acoustic implants, which are designed to treat a range of moderate to profound types of hearing loss.
Over 450,000 people of all ages, across more than 100 countries, now hear because of Cochlear.
# # #
References:
- Data on file - Hi-Focus Mid-Scala Electrode brochure (028-M270-03). https://www.advancedbionics.com/content/dam/ab/Global/en_ce/documents/professional/HiFocusMid-Scala_Electrode_Brochure.pdf and Flex 2.
- Cochlear Limited recipients registered with Nucleus Profile cochlear implants, August 2016.
- Data on file - CLTD5446: Clinical investigation of the Nucleus CI532 cochlear implant.
- Data on file - CI532 Temporal Bone Usability Test Report, 2014.
- Holden LK, Finley CC, Firszt JB, Holden TA, Brenner C, Potts LG, et al. Factors affecting open-set word recognition in adults with cochlear implants. Ear Hear.2013 May-Jun; 34(3):342-60.
- Esquia (2013): Esquia Medina, GN., Borel, S., Nguyen, Y., Ambert-Dahan, E., Ferrary, E., Sterkers, O., Bozorg Grayeli, A. Is Electrode-Modiolus Distance a Prognostic Factor for Hearing Performances after Cochlear Implant Surgery?. Audiol Neurotol 2013;18:406–413. DOI: 10.1159/000354115.
- Van der Beek (2005): van der Beek, FB., Boermans, PP., Verbist, BM., Briaire, JJ., Frijns, JH. Clinical evaluation of the Clarion CII HiFocus 1 with and without positioner. Ear & Hear. 2005 Dec;26(6):577-92.
- Dowell, R. Evidence about the effectiveness of cochlear implants for adults. Evidence based practice in audiology: evaluating interventions for children and adults with hearing impairment. Plural Publishing. 141-166. 2012.
- 588021 | Data on file - EA32 Electrode Insertion Safety and Performance Study, 2015.
- Data on file - EMEA CI532 FEP 2015 - D806144.